Drug development is a complex journey from discovery in the laboratory to availability on pharmacy shelves. Central to this process are two critical regulatory steps of the ind vs nda: the Investigational New Drug (IND) application and the New Drug Application (NDA). These applications are integral to ensuring the safety and efficacy of new pharmaceuticals. The IND serves as a gateway, allowing researchers to conduct clinical trials that are essential for assessing a drug’s effects on humans. Following successful trials and comprehensive data collection, the NDA marks the next crucial phase. This application aims to demonstrate a drug’s safety, efficacy, and quality to regulatory bodies, primarily the U.S. Food and Drug Administration (FDA). Understanding these steps is vital for pharmaceutical companies striving to bring new drugs to market while maintaining compliance with stringent regulatory standards.
Purpose of IND and NDA in Pharma
The Role of the IND in Drug Development
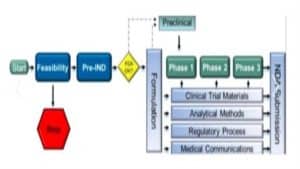
The Investigational New Drug (IND) application is pivotal in the early stages of drug development. It represents the first major interaction between pharmaceutical companies and regulatory authorities. The primary function of the IND is to secure authorization to commence human clinical trials. This step is crucial because it ensures that a drug candidate can be administered safely to humans for the first time. An IND application comprises several components, including preclinical study results, manufacturing information, and clinical trial protocols. Preclinical studies provide foundational data about a drug’s safety profile, which is necessary before human testing. Manufacturing information describes the composition, manufacturing processes, and controls to ensure consistent production quality. Clinical trial protocols outline the proposed studies, offering insights into study design, methodology, and informed consent procedures. Regulatory agencies meticulously review these details to assess potential risks and benefits.
Clinical Trials and Their Importance
Once an IND is approved, the focus shifts to clinical trials, which are conducted in a series of phases. These trials are designed to evaluate the safety and efficacy of the drug in human subjects. Phase I trials are primarily concerned with safety, determining a drug’s pharmacokinetics and pharmacodynamics. This involves small groups of healthy volunteers or patients and aims to identify side effects and establish safe dosage ranges. Phase II trials involve a larger group of participants and focus on the drug’s efficacy while continuing to monitor safety aspects. These trials determine whether the drug has the intended therapeutic effect and hone in on the optimal dose. In Phase III trials, the drug is tested on an even larger population to confirm its effectiveness, monitor side effects, and compare it against standard treatments. This phase gathers the comprehensive data needed to advance to an NDA submission. Each clinical trial phase is crucial in building a robust data set that supports the drug’s safety and efficacy profile, which will be critical during the NDA review process.
Preparing the NDA Submission
After clinical trials successfully demonstrate a drug’s safety and efficacy, the next step is preparing the New Drug Application (NDA). This submission is a comprehensive document that compiles all data pertaining to the drug, encompassing everything from preclinical research and clinical trials to manufacturing specifics and proposed labeling. The NDA must provide compelling evidence that the drug is safe and effective for its intended use. It should also assure the regulatory body that the manufacturing processes are well-controlled and consistent. A critical component of the NDA is the integration of clinical data, which requires a meticulous analysis of trial results to substantiate claims of safety and efficacy. Another important aspect is the proposed labeling, which should clearly communicate the drug’s benefits, risks, and usage instructions to both healthcare professionals and consumers.
Conclusion
Navigating the IND and NDA processes presents numerous challenges. Regulatory requirements are stringent, demanding thorough documentation and data integrity. Companies often face hurdles in aligning preclinical and clinical data with regulatory expectations, necessitating clear strategic planning and expert guidance. To succeed, it’s vital to establish open communication channels with regulatory bodies from the outset. Engaging skilled regulatory affairs professionals can facilitate smooth interactions and help anticipate potential issues.


